The Cold Facts About a Hot Commodity: LNG
Posted by aeberman on October 4, 2012 - 8:35am
This is a guest post by Bill White. Bill is a Researcher/Writer, the Office of the Federal Coordinator for Alaska Natural Gas Transportation Projects (OFC).

Photo courtesy of ConocoPhillips
Liquefied natural gas is an odorless, colorless, non-toxic, non-corrosive and non-flammable form of methane. As fuels go, it's pretty cool.
Actually, LNG is colder than Antarctica on winter solstice. Methane is chilled to about minus 260 degrees fahrenheit — a temperature that transforms it from a vapor to a liquid, compressing its volume 600 times to make it more economical to store for later use or to ship long distances from countries endowed with natural gas to those starved for the fuel.
That's the broad story of LNG — a case of Adam Smith capitalism at work.
But in the details, the LNG story is a tale of brilliant physicists, savvy government engineers and entrepreneurial risk takers. LNG's back story includes a Nobel Prize, anxiety about U.S. air defense and a disaster that destroyed part of Cleveland.
LNG touches only a small portion of the world's gas supply, but it's the fastest-growing portion. Since 2000, global demand for LNG has grown 140 percent and now accounts for roughly 10 percent of the methane consumed worldwide. The rest moves to market by pipeline.
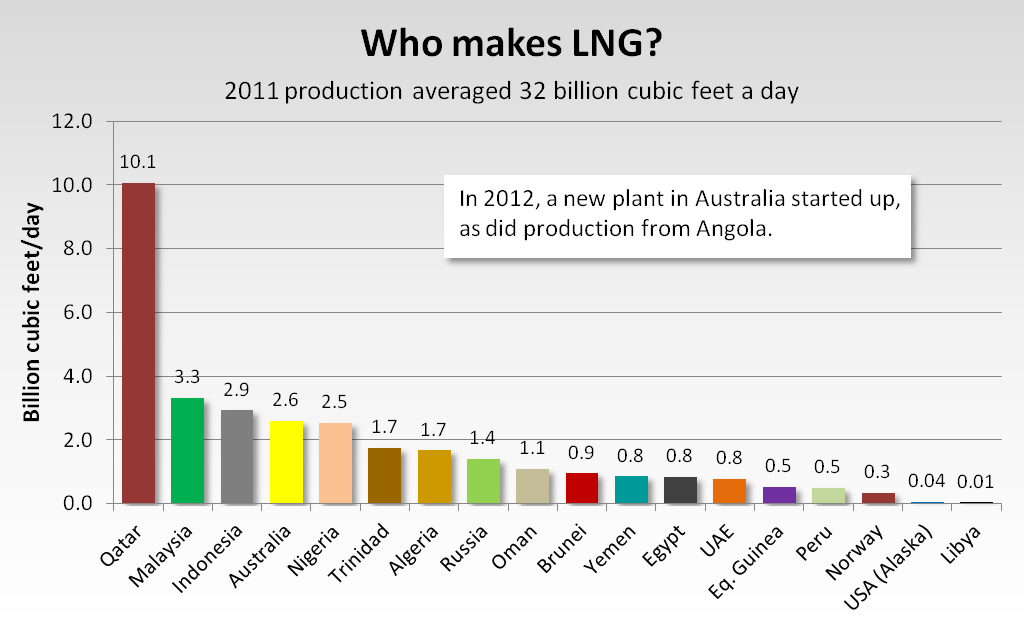
LNG is exported from 19 countries, including from one U.S. plant in Nikiski, Alaska.
Since 2006, Norway, Russia, Yemen, Peru, Angola and Equatorial Guinea all have started making LNG, while Qatar, Nigeria, Australia, Oman and Indonesia have expanded production.
Qatar's expansion was an act of sheer audacity. Qatar tripled its LNG production capacity to over 80 million metric tons a year — about 11 billion cubic feet a day — leaping past Malaysia and Indonesia as the world's largest LNG maker. Last year Qatari plants exported almost one-third of the LNG traded across the globe. In the mid-2000s, with construction under way, Qatari officials thought they'd be selling much of their LNG to the United States. The Lower 48 shale-gas boom blew apart that plan. But last year, as Japan idled nuclear power production after the Fukushima disaster, Qatari exports to Japan soared 56 percent over their 2010 level, according to the BP Statistical Review of World Energy [1]. That dulled Qatar's pain of losing the U.S. market.
Meanwhile, more countries are clamoring for LNG to quench their growing energy appetite.
Since 2006, China, Brazil, Chile, Dubai, Kuwait, the Netherlands and even Canada and Mexico all became first-time importers of LNG. They joined the mainstay LNG consumers of Japan, South Korea and Taiwan, according to the International Group of Liquefied Natural Gas Importers [2].
In all, 25 countries took LNG shipments last year, the gas importers group said.
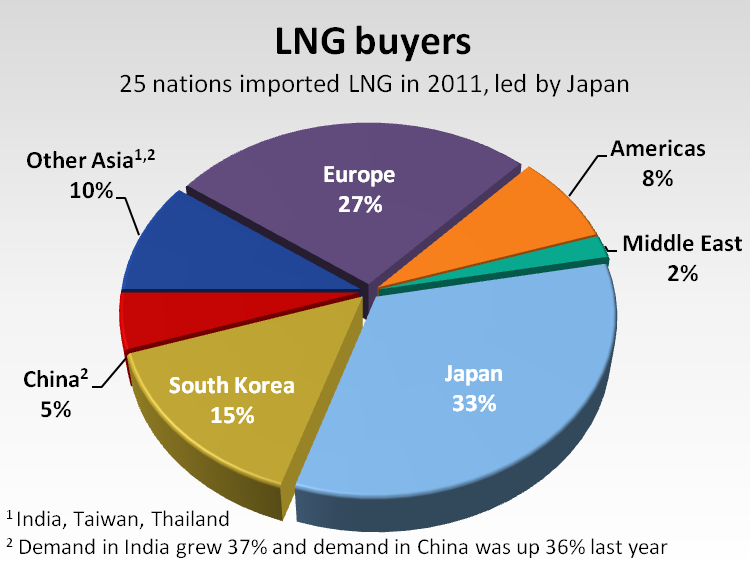
As the world's demand for LNG grows, more locations are mulling entry into the production game. Export projects in Western Canada [7], Eastern Africa, Russia and the U.S. Gulf Coast are under consideration.
One other possible contender: Export of LNG made from Alaska North Slope gas. The main North Slope producers — ExxonMobil, ConocoPhillips and BP — jointly are at an early stage [8] of considering such a project.
HOW IT WORKS
Chemical engineers have known for years how to liquefy vaporous methane.
And for decades LNG tankers — essentially massive thermos bottles that keep the gas cold and liquid — have sailed the oceans safely.
Like many great inventions, liquefied natural gas [3] emerged as an industry via a progression of events over many years, responding to both commercial and geopolitical pressures.
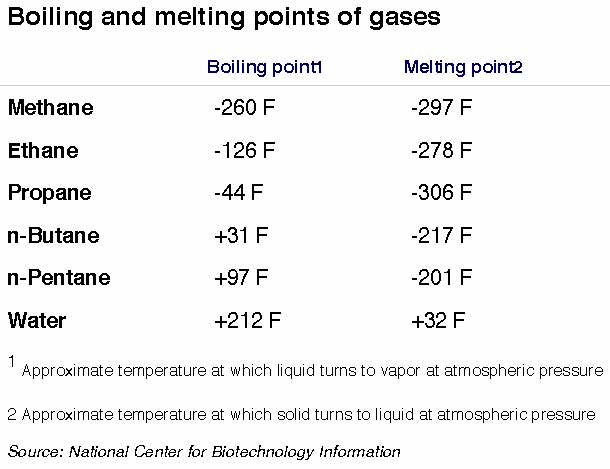
A key development was learning methane's "boiling point," a temperature below which methane is a liquid and above which it's a vapor.
Most people likely are familiar with the boiling point of water: 212 degrees. Heat water above that temperature and the liquid becomes a vapor.
Methane's boiling point is about minus 260 degrees. Above that frigid temperature it's a vapor. Below it and you have a liquid.
But liquefying natural gas involves more than superchilling it and maintaining the temperature.
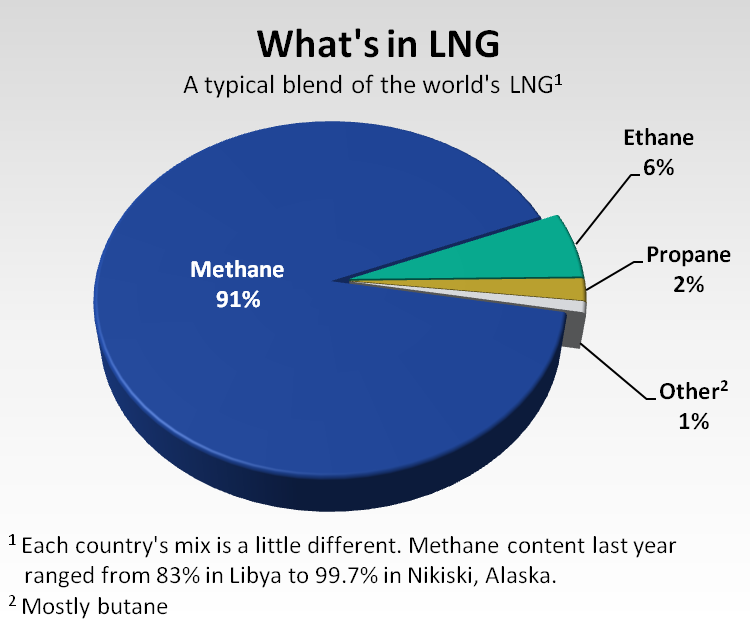
That's because a natural gas stream rising out of the ground contains more than just methane, although methane usually is the main component. The ethane, propane, butane, pentane, carbon dioxide, water and other components each have separate boiling points.
Ethane liquefies at minus 127, propane at minus 44, butane at plus 31 degrees, and so on. Like water at 32 degrees, these gases also have "melting points," a temperature below which they become solid. (Dry ice is nothing more than solid carbon dioxide, whose melting point is minus 109.)
These gases have different boiling and melting points because although they're all hydrocarbons — composed of hydrogen and carbon atoms — the number of atoms differs. The more carbon atoms a molecule contains, the heavier it is. That weight determines the temperatures and pressures that make the gas a vapor or liquid.
Methane has the fewest carbon atoms — one — so it has the coldest boiling point of these gases. If the entire produced natural gas stream were liquefied, some components — such as butane with its four carbon atoms and pentane with its five — would freeze solid before the methane vapors got cold enough to become liquid.
Chilling the entire gas stream to minus 260 to liquefy methane thus could produce a slushy slurry of product that would muck up the machinery. This is why the heavier hydrocarbons mostly are stripped from the gas stream before liquefaction.
THE PROCESS
Here's a quick walk along the LNG value chain:
Step one: Clean the natural gas stream so that mostly methane is being processed. The residual ethane and other components left behind after processing are in quantities too small to matter.
Sometimes this cleansing occurs before the gas reaches the liquefaction plant. More typically cleansing occurs at the plant.
Buyers in Japan and Europe typically like their LNG to be spiked with a little ethane or other carbon-rich gases because their mainstream gas burns hotter than mainstream gas in North America. Ethane, propane, butane, etc., have higher Btu contents than methane and serve as the spiking agents.
Step two: Superchill the methane.
A variety of techniques will liquefy methane. A Pennsylvania company called Air Products [4] licenses the technology that dominates the industry.
Air Products uses several variations on the same process. Essentially, it starts by using propane to precool the methane. Propane is compressed and condensed, then its pressure is eased in steps to provide refrigeration that cools the methane. (Gas warms as it is compressed and then cools as the compression eases. This principle is applied throughout a typical liquefaction process.)
Next, the cooled methane enters the main stage, a heat exchanger where the gas comes in contact with a blend of refrigerants that transforms the methane vapor into a liquid. Air conditioners work in a similar way: warm air passes over coiled tubing filled with a cold gas.
A new variation uses nitrogen as a final superchilling refrigerant. This allowed much bigger LNG plants to get built, and it partly explains how Qatar could construct so much capacity in recent years.
A technology that's a distant second in the market to Air Products' is licensed by ConocoPhillips. The company's Nikiski, Alaska, plant as well as plants in Trinidad and Tobago, Egypt, Angola, Equatorial Guinea and one site in Australia use it.
ConocoPhillips routes cleansed methane first into a propane heat exchanger to initially drop the temperature. Ethylene is used to drop the temperature more (you can make ethane colder than propane before it boils into a vapor). Then the gas enters a methane cold box connected to mighty compressors to cool the gas to near a liquid state. A final "flash blast" finishes the job.
Most LNG plants have on site more than one processing unit — called trains. The trains operate independent of each other, running in parallel to liquefy methane. Qatar hosts the world's largest trains — the biggest can handle about 1 billion cubic feet of natural gas per day. Qatar's most massive plant, at the Ras Laffan complex, features two such trains plus four smaller ones that together can process about 5 bcf a day. That's about twice the volume as has been discussed for an LNG plant that could process Alaska North Slope gas. Alaska's Nikiski plant is relatively small, with capacity to handle about 200 million cubic feet a day.
One final point about liquefying methane: About 10 to 15 percent of the gas gets consumed during the process. Much of it to run the plant's turbines, compressors and other machinery.
Step three: Store the LNG until it's shipped to market. Special insulated metal tanks keep the gas liquid. A small fraction will "boil off" — warm into a vapor — and this gas can be reliquefied or used to power the plant.
Storage tank dimensions vary widely, depending on whether the LNG is stored for truck fueling, peak shaving or import-export. The largest storage tanks stand as tall as a 14-story building (about 170 feet tall), are nearly as wide as a football-field length (280 feet in diameter) and can hold up to 200,000 cubic meters of LNG — the equivalent of roughly 4 billion cubic feet of vaporous methane, or about one-15th of daily U.S. gas production last year. In short: They can be big.
Step four: Ship the gas. Special tankers with insulated chambers keep the gas below minus 260. Again, a small volume of liquid methane vaporizes on the trip to market; this gas typically is used to power the ship or is reliquefied.
At the end of 2011, 360 ships comprised the global LNG fleet, according to the International Gas Union [5]. Ships typically get built in tandem with LNG plants and get contracted to sail between the plant and its customers. Just as the capacity to make LNG has skyrocketed in recent years, so has the tanker capacity, growing 150 percent since 2006, the IGU said.
The average tanker capacity is about 3.1 bcf of gas (after the liquid gets converted back into a vapor). South Korea is the big builder of tankers. An average one can cost at least $150 million. The largest tankers were built for the Qatar expansion. They can carry about 5.5 bcf, but the tankers are too big for some LNG receiving ports.
Step five: Convert the liquid back into a vapor, called regasification.
This happens in the LNG destination port. LNG is offloaded into storage tanks. The LNG then is warmed into vapor as needed before entering the local gas pipeline system.
THE CRYOGENICS CRAZE
As an export product, LNG dates back less than 50 years, to 1964.
That year, as Ford rolled out its new sports car, the Mustang, a British shipyard launched the Methane Princess, a tanker that carried the first commercial load of LNG, from a new plant in Algeria to a gas-hungry United Kingdom.
Within a few years, Algeria was sending LNG to France, too, and Libya was exporting it to Italy and Spain. In 1969, a new Phillips and Marathon plant in Nikiski, Alaska, started shipping LNG made of Cook Inlet natural gas to Japan, inaugurating LNG trade to Asia. Japan is the world's top LNG consumer today.
But the true history of LNG dates to 100 years earlier as scientists studied how very low temperatures changed matter, a specialty called cryogenics.

In the 1870s, German engineer Carl von Linde's pioneering work in compressed refrigeration found a ready market among breweries and slaughterhouses. Von Linde's technique for chilling air to extract the oxygen, developed around the turn of the century, also was a transforming moment. Isolating oxygen led to development of a torch that revolutionized metal cutting as well as welding for skyscrapers.
Other scientists and engineers hopped aboard the cryogenics craze.
Ethane for plastics, chlorine for sanitizing sewage, oxygen for hospital patients, nitrogen for cryosurgery are among the thousands of products and uses that trace their origins to chilling gases to isolate their components.
THE GAS THAT WOULDN'T BURN
The birth of liquefied methane stemmed from work that used cryogenics to isolate helium.
Helium is a marvelous gas that has been adapted to many uses [6] today, such as cooling superconducting magnets in medical MRI scanners.
If helium isolation has a Eureka! moment, it arguably is a 1903 event [7] in a small flatland town called Dexter, Kan.
A driller hit a "howling gasser" of a well there. Nine million cubic feet of gas spit to the surface each day before the well could be capped. Dreams of riches infused the locals. Ore smelters. Brick and glass plants. Soon they would be wildly prosperous.
To celebrate, the town tossed a huge party, the climax of which was to be lighting the gas jet. After speeches, a bale of burning hay was nudged to the escaping gas to produce a promised "great pillar of flame." But the gas failed to ignite. To everyone's surprise, the burning bale got snuffed instead.
A geologist and a chemistry professor soon teamed to solve the mystery of the gas that wouldn't burn.
They discovered the gas was mostly nitrogen. The amount of methane present wasn't enough to combust given all the non-flammable nitrogen — just as trace quantities of methane in the Earth's air don't burst into flame every time someone lights a cigarette.
They also found "inert residue" present in the Dexter gas. After further analysis, they learned this residue included helium.
This discovery was astonishing. To that time, helium was considered a rare element. But now it seemed helium could be found in an ordinary natural gas stream. As for Dexter, it was located in the planet's great cradle of helium: The natural gas deposits of the U.S. plains.

The scene then shifted to the lab of Dutch physicist Heike Kamerlingh Onnes. In 1908, he was the first to liquefy helium, chilling helium through a series of stages until getting it to minus 452 degrees, at which point the vaporous helium transformed into liquid helium, reaching its boiling point. It was the coldest temperature ever achieved on Earth. Onnes won the Nobel Prize in Physics [8] five years later for his work.
World War I, with cryogenic isolation, became the great leap forward for helium and led eventually to the liquefaction of methane.
During the war, airships — dirigibles, zeppelins and the like — became an novel innovation of combat [9]. Germans dropped bombs from them. The British hunted U-boats. A downside was hydrogen, the lighter-than-air gas used to float most airships. Hydrogen is spectacularly flammable, as the famous 1937 Hindenburg disaster [10] demonstrated.
But helium isn't flammable. The U.S. launched a crash research program in 1917, as the country entered World War I, to find cheap ways to extract large volumes of helium from natural gas and stockpile it.
This research led the U.S. Bureau of Mines in 1924 to produce the first liquid methane as a byproduct of helium separation.
LNG'S EARLY YEARS
During the ensuing years, techniques for liquefying methane were refined and ideas for storing and transporting LNG were patented.
A public revulsion toward flaring natural gas as a waste product of oil production helped propel the industry. Better ways had to be found to move the gas from where it was produced and not needed to where it could be used. The solutions included long-distance pipelines for domestic transport and, much later, LNG for cross-ocean transport.
By 1941, science and capitalism converged to make commercial use of LNG.
That year the East Ohio Gas Co. built a plant in Cleveland that could process about 4 million cubic feet of gas per day into LNG. The company installed three insulated storage tanks to keep the LNG cold. The gas utility regasified LNG when customer demand peaked during winter.
This "peak shaving" concept is a key function of LNG today, the little publicized cousin of making large quantities of LNG for export. Small peak-shaving liquefaction plants [11] and storage sites exist across the world.
The Cleveland operation ran smoothly for three years, until 1944 when the utility installed a fourth storage tank. It was war time, and many metals were in scarce supply for civilian use. The metals on this tank were inferior and failed on Oct. 20, 1944.
An estimated 1.2 million gallons of LNG spilled, so much that it flowed over the protective dike.
The liquid spread like batter on a griddle. Some dropped into the sewers, which filled with methane vapor as the LNG warmed above methane's boiling point. Gas seeped into basements. Houses blew apart as the gas contacted hot-water heater pilot lights.
The Cleveland catastrophe killed 128 people; 14,000 became homeless.
The LNG industry went dormant, except for a liquefaction plant Dresser Industries built for the Soviet Union in 1947.
HEADED TO SEA

The idea of water-borne LNG deliveries started to get traction in the mid-1950s.
A joint venture of Continental Oil Co. (Conoco) and Union Stock and Transit Co., a Chicago stockyards operation, did pivotal work on how this idea could work. The venture's name was Constock, a blend of the partners' names.
Union originally wanted Gulf Coast methane barged as LNG to Chicago for refrigeration at its slaughterhouses. But in the late 1950s, with the United Kingdom, Japan and other countries expressing interest in LNG, the focus turned to trans-ocean shipments.
Constock worked on designing the entire system, from liquefaction to regasification. In 1959, a test shipment of LNG left a new plant near Lake Charles, La., and sailed to a new receiving terminal on Canvey Island, down river from London. The ship — and its LNG cargo — weathered the rough Atlantic well. More test shipments ensued, proving that international trade of LNG could work.
New gas discoveries in Algeria made that country the first mover in LNG exports. The Methane Princess, carrying the world's first commercial load to Canvey Island, was small by today's standards. It could carry up to about 500 million cubic feet of gas (after regasification). The average LNG tanker today is five times larger.
But the Methane Princess proved to be a workhorse through the early years of LNG export. The vessel was finally scrapped in India during the mid-1990s. Another tanker with the same name sails in the LNG trade today.
---------------------------------------------------------------------------------------------------------------------
More Pipeline Topics
LNG industry says today’s operations are safe
As tragic as the Cleveland disaster was, it did imbue the LNG industry with a culture of safety.
If you’ll give them time, people from the industry will talk endlessly about safety within the entire LNG value chain, from liquefaction to storage, tankers and regasification. These operations are heavily regulated [12] for safety across the world, and industry members will even boast [13] about that regulation and insist that potential hazards are manageable.
To illustrate the concept of safety, visitors to ConocoPhillips’ plant in Nikiski see a series of demonstrations aimed to demystify LNG, including:
A plant manager pours LNG on the floor. Instantly, the gas forms into clear beads then — poof — vaporizes as it warms while absorbing heat from the carpet and air.
The manager dunks graham crackers in LNG then invites guests to eat them. They do so warily, misty vapor wafting from their mouths as they chew. This stunt can be an acutely effective in LNG-leery towns when the people consuming the crackers are children of community leaders and opponents.
The LNG industry does have a strong safety record, marred mainly by the Cleveland disaster, a fire and death at a Maryland import plant in 1979 and an explosion that killed 27 people at an Algeria liquefaction plant in 2004.
As an industry website [14] puts it: “LNG is transported many miles as it crosses the ocean, transferred to storage tanks, converted back to natural gas and then sent to market. The LNG industry has spent a considerable amount of time analyzing and assessing the hazards along the way and has either eliminated or developed mitigation techniques to reduce risks. As a result, in more than 50 years of commercial LNG use, no major accidents or safety or security problems have occurred, either in port or at sea.” (The Maryland accident actually was 33 years ago.)
The site stresses that “LNG is not explosive.” But the vapors are flammable — if they comprise 5 to 15 percent of the air and something ignites them. U.S. regulations require safety zones around LNG facilities so that any vapors accidentally released get fully diluted before they reach the property line.
University of Texas researchers [15] concluded that although “LNG operations are industrial activities,” LNG can be safely transported and used if regulators hold the industry to the safety standards and protocols that have developed over time.
------------------------------------------------------------------------------------------
Source URL: http://www.arcticgas.gov/lng-cold-facts-about-hot-commodity
Links:
[1] http://www.bp.com/sectionbodycopy.do?categoryId=7500&contentId=7068481
[2] http://www.giignl.org/fileadmin/user_upload/pdf/A_PUBLIC_INFORMATION/LNG...
[3] http://www.giignl.org/fileadmin/user_upload/pdf/A_PUBLIC_INFORMATION/LNG...
[4] http://www.airproducts.com/
[5] http://www.igu.org/igu-publications/LNG%20Report%202011.pdf
[6] http://physicsworld.com/cws/article/news/2010/jan/27/helium-sell-off-ris...
[7] http://acswebcontent.acs.org/landmarks/landmarks/helium/helium.html
[8] http://www.nobelprize.org/nobel_prizes/physics/laureates/1913/
[9] http://www.worldwar1.com/sfzepp.htm
[10] http://www.airships.net/hindenburg/disaster
[11] http://www.arcticgas.gov/sites/default/files/documents/2003-eia-lng-mark...
[12] http://www.energy.ca.gov/lng/safety.html
[13] http://www.netl.doe.gov/publications/proceedings/02/ngt/Quillen.pdf%20
[14] http://www.lngfacts.org/About-LNG/Safety.asp
[15] http://www.beg.utexas.edu/energyecon/lng/documents/CEE_LNG_Safety_and_Se...



Apart from being one of the most effective greenhouse gasses there is when released.
Bill... great overall article on LNG.
Here were I live, UPS is running 36 line-haul trucks on LNG. I believe the US Govt gave them $4 million dollars for the purchase of the trucks. I spoke with several of the drivers and they tell me that the biggest drawback is the constant refueling.
The trucks drive from Beaver, Utah to Las Vegas, Nevada and back. The night shift runs up to Salt Lake, Utah and then back to Beaver to complete the cycle. When they were driving diesel trucks one fueling (200 gallons) would allow them to make nearly a complete round trip. Now, they have to fuel at each stop. That's four refuelings a day.
Why? Because one gallon of diesel equals 1.7 gallons of LNG. They are running a 95% LNG to 5% diesel system. Their total LNG capacity is 190 LNG gallons... which is really 111 gallons of diesel gallon equivalent (DGE).
When they first started the new LNG trucks, some would run out of fuel before they made it back to Beaver, Utah. This was a bummer because there were no LNG refueling stations between Beaver and Las Vegas. So, now they have to refuel at every stop just to make sure there is enough fuel.
Another interesting aspect, is fueling with LNG... a person has to be certified. The extreme low temperature of -260 degrees can cause skin burns and etc. The Blu LNG station in Beaver, Utah is open 24 hours a day and has at least one certified fueling person (two during peak hours).
These certified attendants wear a helmet with large plastic face guard to protect against spashing. This is nothing like fueling regular old diesel.
Typically a LNG truck will cost between $30,000-$100,000 more than a typical diesel semi-tractor.
There are plans to open LNG filling stations across the USA to get more large trucks on LNG. This is a good idea on paper, but when you start to realize that stated reserves of shale gas have been severaly overstated, this LNG infrastructure may indeed be too little too late.
Lastly, the mileage these UPS trucks are averaging is about 5-5.2 mpg on gallon of DGE of LNG. It turns out to be about 8-10% less than typical diesel engines.
Again, I believe LNG is a good alternative source for diesel, but I don't believe there is a long term growth supply of natural gas to run the interstate trucking system.
As a truck fuel it sounds idiotic and pure politics. Why not just use propane or butane at ambient temperature?
Propane liquifies at -44 f. Trucks using propane would have to avoid the northerly states. Other than that, it seems that a lower heat might be better in the South, though that also lowers the effective energy.
With less carbon than NG, perhaps LNG improves AGW impact a bit over propane or butane. Still in all, it still releases CO2 when burned, and isn't that something that we should be avoiding?
Craig
Just for the record, cold weather isn't an issue for propane powered trucks.
They use heated engine coolant to assist liquid fuel vaporization.
Operating Temperature Range ………………-40° C to 120° C (-40° F to 250° F)
Thanks. Not sure this would be low enough for some, though. Of course, diesel fuel coaglates in extreme cold before propane liquifies (I sat for several days waiting for temps to rise so my diesel would start. Even with a motor block heater!
Big rigs that travel in higher latitudes have to keep 'em running!
Craig
indeed , why not NG as a gas ?
http://www.lowtechmagazine.com/2011/11/gas-bag-vehicles.html
and ofcourse modern public transfport
http://dfw.cbslocal.com/2011/01/12/dart-buying-compressed-natural-gas-po...
but for the big American tractor/trailers I can see why you'd want to stay with diesel
Forbin
According to CleanEnergyFuels 2011 Annual report, medium and large trucks consumed 36 billion gallons of diesel in 2011. To switch this amount over to LNG, this would be the conversion:
1 gallon low sulfur diesel = 130,000 BTU's (rounded)
1,000 cubic feet natural gas = 1 million BTU's
36 billion gallons X 130,000 BTU = 4.68e+15 BTU'S
4.68E+15 BTU's = 4.68 trillion cubic feet natural gas
However, according to Bill's article, 5-10% of the natural gas is consumed in the process, so if we add an additonal 10% it would take over 5 trillion cubic feet per year to get the medium and large sized trucks totally on LNG.
We may get a small percentage on LNG, but I highly doubt we will have the natural gas resouces to be able to make Boone Pickens plan work.
I wonder how much energy is required to compress CNG in comparison to the 10% that's needed to liquify LNG. I googled but didn't find anything.
From what I have been told CNG takes from 2% to 6% to compress. This varies depending on the pressure used. Higher pressures mean smaller tanks, more compression energy, and heavier tanks. Just some of the tradeoffs. LNG is not practical for autos, because of the fact they may be stored in enclosed spaces for unlimited times giving rise to gas venting as the tanks heat. This is not a problem with commercial trucks.
4.68 trillion cubic feet natural gas ... 5-10% of the natural gas is consumed ...
That's less than 18% of current US gas production. With the industry idling rigs given the low price, I don't see the required qty of gas as being any great problem. Deploying a retooled LNG truck fleet and distributing LNG to every truck stop is another matter.
In Asia, public transport is switching to NG really fast, even if local demand is stalling:
http://asie-info.fr/2012/09/28/malaisie-des-bus-pourpres-qui-roulent-ver...
Propane is as expensive as diesel, per BTU.
Typically a LNG truck will cost between $30,000-$100,000 more than a typical diesel semi-tractor.
So what's the price of LNG at the pump?
The market price of gas (not liquefied) is $3/million BTU. If LNG at the pump is ~$4/million BTU and diesel is 130K BTU per $4 gallon or $31/million BTU, then diesel is 7.5 times more expensive per unit energy. At 200 gallons of diesel a day, the savings would be almost $700/day for the LNG rigs. The extra $100K invested in the LNG truck would pay for itself in less than a year.
Sound right? If so I'd pay the drivers what ever they wanted to lessen their annoyance at topping off LNG rigs.
Modifications for Methane injection on diesel engines are relatively modest. Most of this cost is probably for documentation, testing, and of course the normal bloat of a government funded project.
the LNG tanks are very expensive. From the data I have seen, one LNG tank for a big rig is $10,000.
The Blu LNG filling station in Beaver, Utah has a gallon (DGE) of LNG at $2.65.
LNG at $2.65/gal is $34/million BTU, *more* expensive than diesel at $4/gallon. What a waste.
Falstaff... that might be, but Clean Energy Fuels isn't making any money on producing LNG & CNG yet. They are one of the largest producers of LNG in the U.S. If we look at the 2011 Annual Report, they stated the following:
2011 Total Revenues = $293 million
2011 Total Operating Expenses = $331 million
2011 Operating Loss = $38.5 million
Production delivered to market
2011 CNG gallons = 101.8 million
2011 LNG gallons = 47.1 million
Those figures are based on equivalent gasoline gallons. So, even though Clean Energy Fuels can create 7 gallons of LNG (diesel equivalent gallons)from one mmbtu natural gas contract which is currently $3.42, they are still losing money.
In 2011, their cost of sales was a staggering 75% of their total revenue. I gather, once they ramp up production significantly, their cost of sales in percentage terms will decline.
even though Clean Energy Fuels can create 7 gallons of LNG (diesel equivalent gallons)from one mmbtu natural gas
Well that's the question. Theoretically that's possible but, practically, can they? Or perhaps they have a long term gas contract going back to $12/mmbtu gas.
Waste Management is embracing Natural gas.
Source
LNG: What could possibly go wrong?
http://en.wikipedia.org/wiki/Port_of_Boston
http://www.chelsearecord.com/2010/01/14/chelsea-boston-harbor-and-lng/
http://www.reuters.com/article/2011/10/26/us-lng-cheniere-idUSTRE79P3DO2...
Best hopes in Boston!
Craig
I'm confused...I think here in town we have several pumps that have CNG...what is the difference? One uses high pressure, one use cold temps?
LNG is basically at atmospheric pressure, just slightly above. It's kept as liquid by temperature, not pressure. CNG is obviously at high pressure. LNG tankers and storage tanks are basically just giant thermos bottles. CNG scares me more than LNG. You wouldn't have CNG pumps, they would be compressors. LNG would be pumps, and they would have to be cryogenic.
daddylonglegs, I wondered the same thing you were asking. I had only heard of the compressed natural gas for fueling vehicles, however I guess there is liquid natural gas too.
Yes, apparently there are cars that have little LNG coolers in their trunks.
I had not realized anyone was using LNG as a motor fuel source. From:
http://www.consumerenergycenter.org/transportation/afvs/lng.html
California's LNG fuel infrastructure is expanding rapidly. By the end of 2005, there will be more than 40 locations strategically located near major highways and thoroughfares throughout the state. To further enhance the supply and distribution, there are plans to construct one or more fueling terminals in Southern California and/or Mexico.
An average LNG fueling station costs about $1 million. These locations store and dispense fuel to vehicles.
Through 2005, Energy Commission programs have provided more than $3.5 million in grant cost-share funding for 15 infrastructure projects. The total cost of these projects is more than $20 million.
Because of LNG's increased driving range, it is used in heavy-duty vehicles, typically vehicles that are classified as "Class 8" (33,000 - 80,000 pounds, gross vehicle weight).
Typical transportation applications are refuse haulers, local delivery (grocery trucks), and transit buses.
Rockman... it looks like the trucks moving the containers from the ports in California are switching more to LNG due to the much lower carbon emissions. Again, as I noted in the comment above, UPS has 36 trucks running LNG and plan on adding more if the US GOVT keeps coughing up the bucks to buy the rigs.
I have also heard, that engines running on LNG after 500,000 miles were torn apart to show hardly any wear or carbon deposits in the engine.
I find the logistics for LNG fueling of cars to be very difficult to imagine. Trucks are easier because they tend to have a set schedule and route, and a predictable fuel usage.
If you have a CNG car and park it for the night, nothing happens. Your tank is pressurized but that's about all. You have an LNG tank in your car and you park it for the night - it gets warm and starts vaporizing and building pressure, and building pressure, and building pressure....
ty454... the LNG systems have a relief value that allows the gas to escape so the pressure does not build up. The funny thing, if a truck is allowed to sit for a week, a great deal of the LNG gets released from the tanks. That is why they have to keep moving these trucks... which is the reason why a company like UPS is a great candidate. Their LNG trucks drive 24/7.
Exactly. I wonder where all that vented gas goes while the car is parked in the garage??? Your reasoning is exactly why I think trucks could be set up for LNG but not cars.
One of the coolest things I've seen was looking out of my office window and watching one of the million barrel LNG tanks venting to atmosphere due to a malfunctioned relief valve. It's the weirdest sight. The vapor forms a pure white cloud and just slowly shoots out horizontally about 50ft into the air and just disappears. It's under such a low pressure that it's not violent or anything, but the cloud is just so white, and the way it moves is surreal.
You may be thinking of LPG (liquid petroleum gas, also called Autogas), which is not liquefied methane but liquid propane/butane blend at much higher temperature. It may be refilled by ordinary people without safety gear at many ordinary filling stations in countries where it's popular, and is stored in light vehicles using a lightly-insulated pressure tank like this:
Dispensers have sealed nozzles, mostly for safety purposes. The tank can reach quite high pressures in hot conditions so it's not advised to try filling it until it's really empty.
Thanks for the informative A-Z summary. I am wondering though, if terrorism picks up in the coming years then wouldn't these tankers be sitting ducks? What would happen if the side of one got blown out and all that LNG hits the sea water and boils and explodes at the same time? 1/15th of daily US NG production all blowing up at the same time in the same place?
Null - From the same source above:
Is it flammable? When cold LNG comes in contact with warmer air, it creates a visible vapor cloud from condensed moisture in the air. As it continues to get warmer, the vapor cloud becomes lighter than air and rises. When the vapor mixes with air, it is only flammable when the mixture is between 5-15 percent natural gas. When the mixture is less than 5 percent natural gas it doesn't burn. When the mixture is more than 15 percent natural gas in air, there is not enough oxygen for it to burn.
Is it explosive? As a liquid, LNG is not explosive. LNG vapor will only explode in an enclosed space within the flammable range of 5-15 percent.
A gentle flame:
http://news.yahoo.com/blogs/lookout/wild-gas-tanker-explosion-caught-vid...
Sandia Labs performed just such a study several years ago. You can find it on google. The conclusion was that the risk really wasn't that great. Where you'd run into problems is if liquid was trapped in the hull or in vessel compartments as it vaporized and could bleve due to heat from the attack. But LNG itself is pretty safe in terms of explosions for the reasons that Rockman indicated.
The energy content of a big 135,000M^3 LNG tanker at 22e9J per M^3 is 3e15J, or about thirty times the energy content of the Nagasaki atomic weapon, 9e13J. However, unlike methane, fission does not require air at ~10:1 to release energy.
I suspect an LNG tanker fire and explosion would be on the order of that produced by a gasoline or diesel tanker fire for the reasons addressed above by RockM and others. The LNG has to first come to the boiling point and vaporize which would not happen all at once, then mix with sufficient oxygen that is being depleted by initial combustion so neither would that happen all at once. Since the the unconfined burn can not happen all at once a great deal of the methane must disperse unburned into the atmosphere.
I think the Cleveland leak and fire described in the above article was illustrative: there was no immediate catastrophic explosion. Explosions occurred later when vapor found itself confined in sewers and basements.
What I have in mind is a large missile, or plane, that has in its hold a large amount of liquid oxygen, an ample supply of jet fuel that will detonate and catch on fire on impact with a LNG tanker while spilling the contents of the liquid oxygen into the LNG.
Wouldn't that make for a big psychedelic mushroom cloud?
To get said mushroom cloud, you'd have to figure out how to mix liquid oxygen and liquid methane without the methane freezing.
http://en.wikipedia.org/wiki/ANFO would be far easier to get a mushroom cloud with.
Thanks for the full LNG rundown Bill. I was going ask for some specifics about the current plan to truck 7BCF/year LNG from the North Slope to the two North Pole refineries and the naphtha burning power plant but I found a good analysis of the project included in Ed King's UAF thesis(517KB PDF) first.
It looks like 7BCF/year LNG translates into a little more than a tanker truck each way per hour. The trucks haul 8,500-10,000 gallons of LNG per trip. This translates to roughly 700MF of gas per load or a bit over 27 truckloads a day.
So the the world produces roughly 15 million barrels per day of LNG. In Norway LNG is also beefed up as a type of oil export (?) where total production of liquids is at 1.85 million barrels a day:
"The average daily liquid production in August was: 1 506 000 barrels of oil, 265 000 barrels of NGL and 64 000 barrels of condensate."
http://www.npd.no/en/news/Production-figures/2012/August-2012/
Informative and lively post, so sorry to grumble, and I know this is an US based site, but nothing says the US still has no idea of the rest of the world like a refusal to use Standard Units.
'Water boils at 212 degrees'; only in America.....
Yeah, cubic feet lol
"14-story building , 280 feet in diameter and can hold up to 200,000 cubic meters" did it for me!
NAOM
Yes and how many dozen guineas did it cost....
"The metric system is the tool of the devil! My car gets forty rods to the hogshead and that's the way I likes it." - Grandpa Simpson
I stopped reading when I saw all the imperial measurements. FFS, it makes the site look bad.
Took the words right off my keyboard.
The other issue that needs pointing out is that the author attributes this wonderful initiative to "Adam Smith capitalism", but:
(a) The Cleveland disaster of 1944 occurred because capitalists, acting under the pressure of market imperatives (i.e. shortage of materials) cut corners on quality - precisely as Adam Smith would have anticipated; and
(b) The industry's safety record in the US is, as admitted by the author himself, reliant on stringent government regulation.
It's a small, but chemically pure, example of how someone can be blinded to the truth by their own ideology.
In fairness to an author, I think he has a day job which he would like to keep. He makes it perfectly clear that the 'safety' of LNG is achieved via government regulation, not through free market ideology, and wishful thinking. But he has sprinkled the prose with buzz words that ideologues like to see.
I think the LNG industry is between a rock and a hard place. They do get their capital from people who have capital, but also have an irrational faith in markets and in their own flawed judgement about other peoples safety. They know from bitter experience that without enforced safety practice really bad things really happen. When their financial backers start muttering about operating costs cutting into profits, its nice to blame the costs on regulators rather than on the technology.
In my country, public servants don't have to write political propaganda in order to keep their jobs. When I was a lad at school (back when dinosaurs roamed the Earth), they used to go on about how this was standard practice only for the mob on the other side of the Iron Curtain.
Thermos is a trade name, the proper name is "Dewar Flask".
It seems like a marriage made in heaven; Japan pays top dollar for LNG while struggling countries are willing to sell. That is until the exporter wants to develop their own economy using gas or more likely conserve what they have left.
At Gladstone on Australia's east coast four 'trains' are under construction. LNG tankers and bulk coal carriers will take shortcuts through the Great Barrier Reef with some minor scrapes so far. The gas input to the plant will blend onshore natgas, coal seam gas and any shale gas that materialises. Since the Japanese have paid up to $19 spot price per gigajoule of West Australian gas this east coast gas export facility looks like a bonanza. A tonne of LNG gives about 55 GJ of thermal energy (after expansion) if I recall.
The effect will be that east Australian piped gas will have to match the pre-LNG price at Gladstone. This has clashed with Australia's carbon tax plans. Some 2000 MW of brown coal fired generation was to be closed and presumably replaced with combined cycle gas. However if the east Australian gas price doubles that will be too expensive (nuclear is illegal in Australia) so the buyback plan faltered. The coal operators apparently don't have to give back their $1bn downpayment from the government.
Conclusion...'export land' considerations will apply to LNG, perhaps sooner than we think.
"The effect will be that east Australian piped gas will have to match the pre-LNG price at Gladstone. "
Apparently some Natural Gas retailers are already admitting that electricity can be cheaper than natural gas, even before we have the eastern seaboard natural gas price crunch ...
http://www.climatespectator.com.au/commentary/gas-industry-admits-electr...
So if it plays out as expected, Australia will see a reduced domestic demand for natural gas and increased demand for electricity (ie coal - as you suggested and wind/solar PV).
And what about falling demand? Falling LNG demand is a fact. See the plateau of the 2nd forecast between 2010 and 2015, now we are into it and we can easily affirm the demand will stall for years, in a best case scenario. With stalling demand, normally firms cut investments and production will also fall. You cannot produce what you no longer need, specially whan your reserves and demand forecasts are (were) largely overextimated.

Depends on who the 'you' is. The demand of LNG imports has fallen dramatically in North America, but not everywhere else. LNG everywhere-else demand is still rising as fast or faster than before the shale-gas boom, especially since Fukushima (chart is 2009).
As usual, if Japan could pay more, LNG producers' investments could support a higher production rate. It seems to me, and others, that Japan is paying the gas so much because the LNG cartel are racketeering them because they are in desperate need for energy (after the nuclear mayhem occurred). Please have a look at EIA last report on the more and more expensive Japan's LNG consumption:
see other infos here: http://www.eia.gov/todayinenergy/detail.cfm?id=2430
It seems to me that there are a two distinct types of markets for NG, one where producers and consumer are connected by pipeline and the other where they are not. NG producers that feed pipeline systems are constrained by the demand connected to the pipeline system so for example, in the US while world wide demand for NG is very high, there appears to be very little capacity to serve markets other than those connected to the US pipeline system. I suppose you could refer to US NG production as "stranded" in that respect and it rewards US consumers with low prices while penalizing producers. It appears that a similar situation exists in Europe with respect to the Russian/European pipeline system and the Russian and North Sea producers.
In the other type of NG market, where extensive pipeline systems connecting producers to consumers do not exist, the problem is how to get the product from producer to consumer and that is what LNG is all about. There a couple of issues, one being that LNG processing facilities along with shipping capacity present a bottleneck and the other being that, once the LNG is loaded on to a ship it can then be sold to the highest bidder anywhere in the world. The only constraint is that the buyer must have LNG processing (re-gassification) facilities to handle the cargo.
One interesting aspect of the NG siuation with respect to Japan is that, according to this EIA country analysis of Russia, there are significant NG resources on Sakhalin Island. With far less than 100 miles of ocean separating Japan's Northernmost island from Sakhalin Island, barring issues with earthquakes, it should be economically feasible to ship NG from Sakhalin Island to Japan via pipeline. However due territorial disputes between Japan and Russia dating back to the end of World War II and even centuries before, it is not politically feasible. In the linked article I discovered that:
It is somewhat Ironic that the current diplomatic stand off between Japan and Russian is over the said Kuril Islands. What if Japan were to argue that since Russia now apparently wants control of the Kuril Islands they should give them back "their half" of Sakhalin Island? Of course, we all know that hell's gonna freeze over before that happens. Fascinating Stuff!
The question is, is it really that:
or, is it just markets at work?
The current debate raging in Jamaica over LNG would be assisted by the dissemination of the information in this article and the ensuing comments but so far, my attempts to steer readers of one of the two the main newspapers to the article by mentioning it in comments, has not met with any success. I did suggest in one of the comments I submitted that, the reporters from the newspaper bring themselves up to speed by reading this article so, maybe they'll read it and stop playing as fast and loose with the terms LNG and NG as they have so far. Many articles make the same assumptions that cicerone has made in his post that because NG prices are low in the US, NG prices delivered as LNG should also be low.
Alan from the islands
Earlier this year the guy that sits beside me at work designed a mobile combined-cycle-power-plant-on-an-LNG-barge. Would have been handy after the Japan tsunami.
When the LNG is re-gassified you have a souce of cold, i.e, a refrigerator at near-cryogenic temperatures. What is done with this? Is it a cold sink for a heat engine? or flash freezing food?
The Japanese have used it for freezing fish on an industrial scale. I'm sure there are other applications.
Japanese could use the "LNG cold stuff" to cool down the crippled nuclear installations, now rusting because of salty water used in the first days.
The Australian perspective:
9/5/2012
Queensland plans to export more than 10 times the gas NSW needs (part 3)
http://crudeoilpeak.info/queensland-plans-to-export-more-than-10-times-t...
6/5/2012
Howard's wrong decisions on offshore gas exports start to hit transport sector now
http://crudeoilpeak.info/howards-wrong-decisions-on-offshore-gas-exports...
7/11/2011
Why coal seam gas will not reduce CO2 emissions
http://crudeoilpeak.info/why-coal-seam-gas-will-not-reduce-co2-emissions
13/10/2011
NSW gas as transport fuel. Where are the plans?
http://crudeoilpeak.info/nsw-gas-as-transport-fuel-where-are-the-plans
11/10/2011
Australia's natural gas squandered in LNG exports
http://crudeoilpeak.info/australias-natural-gas-squandered-in-lng-exports
LNG CNG LPG NGL ... I am suffering from alphabet soup. I think that a few posters here and many more people elsewhere get these TLAs confused from time to time.
After 5' on Wikipedia and other links...
LNG = Liquefied natural gas : is natural gas (predominantly methane, CH4) that has been converted to liquid form for ease of storage or transport.
CNG = Compressed natural gas : made by compressing natural gas (NGL), mainly methane.
LPG = Liquefied petroleum gas : prepared by refining petroleum or "wet" natural gas, and is almost entirely derived from fossil fuel sources. Varieties of LPG bought and sold include mixes that are primarily propane (C3H8), primarily butane (C4H10) and, most commonly, mixes including both propane and butane, depending on the season — in winter more propane, in summer more butane
NGL = Natural gas liquids : NGL are naturally occurring elements found in natural gas, and include Propane (alias LPG), butane and ethane, among others. NGL are valuable as separate products and it is therefore profitable to remove them from the natural gas. The liquids are first extracted from the natural gas and later separated into different components.
So is butane a natural gas liquid or a condensate? I think pentane is always a condensate but I could be wrong about this.
Butane is a natural gas plant liquid and also may be a component of natural gas well condensate. The distinction is based on origin and not on composition. Butane goes back and forth between the gas and liquid phases depending on temperature and pressure, which is why they don't like to see too much of it in either natural gas pipelines or gasoline.
Thanks for the historical perspective.
Its always interesting to see how the science and technology was developed and refined over time and the safety lessons learned along the way.
I wonder what lessons we have yet to learn?
How about the lesson about the EROEI of maintaining 17 cows to fill up one tank for your car?!
It might be nice if university students were actually taught to think critically and systemically. Not holding my breath though...
http://www.epa.gov/ord/gems/scinews_cows-per-tank.htm
Emphasis added
https://encrypted-tbn0.gstatic.com/images?q=tbn:ANd9GcTx2QbyNnWbGBUKY5HH...
Dairy cows are maintained to produce milk, not methane.
The methane is wasted now, and reclaiming it is just a bonus, right?
This is a fascinating story about how *government*regulation* enabled the creation of a whole industry worth billions or trillions. I had no idea that government could be so creative. What am I missing?
(tongue firmly in cheek ;-)
I'm puzzled about the LNG fueling of automobiles and small trucks in Calif. The safety of LNG in Nevada/Utah is due to well trained technicians doing all the critical safety steps. This can't possibly happen in an environment where owner-operators of small private vehicles will be wandering about the refueling facility.
Quantity and price go hand in hand. Furthermore, LNG markets are separated in spot and forward markets. Volume on spot markets has been growing versus term markets and is projected to keep growing faster than term.
http://www.nortonrose.com/files/lng-spot-cargo-trading-66332.pdf
Also, regarding explosion danger google a RAND paper written in April 1975 by D.L. Jaquette
Rgds
WeekendPeak